Dicyclomine Hydrochloride IP
Price 2200 INR/ Kilograms
Dicyclomine Hydrochloride IP Specification
- Particle Size
- As per customer requirements (typically 90% passes through 60 mesh)
- Melting Point
- 172C to 176C
- Molecular Formula
- C19H35NO2HCl
- Poisonous
- No (when used as directed in pharmaceuticals)
- HS Code
- 29224990
- Shelf Life
- 5 years Years
- Loss on Drying
- Not more than 0.5%
- Solubility
- Freely soluble in water, ethanol
- Heavy Metal (%)
- Not more than 0.001%
- Smell
- Odorless
- Taste
- Characteristic
- Molecular Weight
- 345.95 g/mol Kilograms (kg)
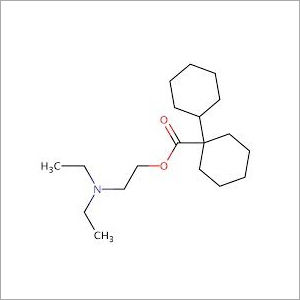
- Structural Formula
- C19H35NO2HCl
- Medicine Name
- Dicyclomine Hydrochloride IP
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- IP (Indian Pharmacopoeia)
- Usage
- Antispasmodic agent in the treatment of irritable bowel syndrome and related conditions
- Purity(%)
- 99% Min
- Physical Form
- Solid
Dicyclomine Hydrochloride IP Trade Information
- Minimum Order Quantity
- 5 Kilograms
- Supply Ability
- 10000 Kilograms Per Month
- Delivery Time
- 15 Days
- Main Domestic Market
- All India
About Dicyclomine Hydrochloride IP
We are famous amongst our customers as a trustworthy organization engaged in offering Dicyclomine Hydrochloride IP. This medications is ideal for treating a certain type of intestinal problem called irritable bowel syndrome. It helps to reduce the symptoms of stomach and intestinal cramping. This medication works by slowing the natural movements of the gut and by relaxing the muscles in the stomach and intestines. Dicyclomine Hydrochloride IP belongs to a class of drugs known as anticholinergics/antispasmodics. It is recommended to take this medication by mouth with or without food.
Further Details:Dicyclomine Hydrochloride IP/BP/USP Dicyclomine Hydrochloride is 2-diethylaminoethyl- bicyclohexyl-1-carboxylate hydrochloride
Formula: C19H35NO2, HCl
Category: Antispasmodic.
Mol. Wt.: 345.95
Dose: 30 to 60 mg daily, in divided doses. If used as an oral solution and the solution is required to be diluted, the diluted solution should be freshly prepared.
| Analysis |
Specification |
| Description |
White or almost white, crystalline powder; odourless or almost odourless. |
| Solubility |
Freely soluble in ethanol (95%) and in chloroform; soluble in water; practically insoluble in ether. |
| Identification: |
|
| A: |
By infra-red absorption spectrum |
| B: |
To 3 ml of a 0.1% w/v solution of sodium dodecyl sulphate, add 5 ml of chloroform and 0.05 ml of a 0.25% w/v solution of methylene blue, mix gently and allow to separate; the chloroform layer is blue. Add 20 mg of the substance being examined dissolved in 2 ml of water, mix gently and allow to separate; the aqueous layer is blue and the chloroform layer is colourless. |
| C: Chloride Test | Dissolve 10 mg in 5 ml of water and add 0.2 ml of 2M nitric acid and 0.5 ml of silver nitrate solution; a white precipitate is produced. |
| D: Melts | Between 172o and 174o. |
| Related Substances |
Any secondary spot in the chromatogram obtained with solution (1) is not more intense than the spot in the chromatogram obtained with solution (2). |
| Sulphated Ash |
Not more than 0.1%. |
| Loss on Drying |
Not more than 1.0%, determined on 1 g by drying in an oven at 105o |
| Assay |
Dicyclomine Hydrochloride contains not less than 99.0 per cent and not more than 101.0 per cent of C19H35NO2,HCl, calculated with reference to the dried substance. |
Superior Quality and Regulatory Compliance
Dicyclomine Hydrochloride IP adheres strictly to Indian Pharmacopoeia standards, ensuring pharma-grade quality with a purity not less than 99%. Rigorously tested for heavy metals and loss on drying, it meets the most stringent quality benchmarks required by global and local markets.
Versatility in Pharmaceutical Formulations
With its excellent solubility in water and ethanol, and customizable particle size, Dicyclomine Hydrochloride IP is readily incorporated into a wide range of dosage forms including tablets, capsules, and syrups. Its characteristic odorless nature and solid physical form further enhance its applicability.
Long Shelf Life and Stability
This API maintains its efficacy and stability for up to five years when stored appropriately. Its solid form, strict control of moisture content, and resistance to contamination make it a reliable ingredient for sustained pharmaceutical manufacturing and distribution.
FAQs of Dicyclomine Hydrochloride IP:
Q: How is Dicyclomine Hydrochloride IP used in pharmaceuticals?
A: Dicyclomine Hydrochloride IP is primarily used as an active pharmaceutical ingredient (API) in the manufacturing of antispasmodic medications. These medicines help relieve muscle spasms in the gastrointestinal tract, commonly prescribed for irritable bowel syndrome (IBS) and related conditions.Q: What is the benefit of using Dicyclomine Hydrochloride IP in formulations?
A: The main advantage is its effectiveness as an antispasmodic agent, offering relief from abdominal pain and discomfort associated with IBS. Its high purity and compliance with IP standards guarantee consistent performance and safety in pharmaceutical products.Q: When should one use medications containing Dicyclomine Hydrochloride IP?
A: Medications formulated with Dicyclomine Hydrochloride IP are typically recommended when a patient experiences symptoms of spasmodic abdominal pain or discomfort due to IBS or similar gastrointestinal disorders. Always use under a healthcare providers guidance.Q: Where is Dicyclomine Hydrochloride IP manufactured and supplied?
A: Dicyclomine Hydrochloride IP is manufactured and exported by pharmaceutical suppliers in India, adhering to strict quality and safety regulations per Indian Pharmacopoeia standards, and is distributed worldwide.Q: What is the process for sourcing Dicyclomine Hydrochloride IP?
A: Pharmaceutical companies can procure Dicyclomine Hydrochloride IP directly from authorized manufacturers and exporters in India. Orders can be tailored to specific particle size requirements and delivered as per customer needs with assured quality and documentation.Q: Is Dicyclomine Hydrochloride IP safe for pharmaceutical use?
A: Yes, Dicyclomine Hydrochloride IP is regarded as non-poisonous when used according to pharmaceutical standards and prescribed dosages. It undergoes stringent quality checks for heavy metals, dryness, and purity, ensuring safety for medicinal applications.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Dicyclomine HCL Category
Dicyclomine Hydrochloride BP
Price 4000 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Solubility : Freely soluble in water and alcohol
Heavy Metal (%) : NMT 0.001%
Type : Other, Active Pharmaceutical Ingredient (API)
Smell : Other, Odorless
Dicyclomine Hydrochloride USP
Price 4000 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Solubility : Freely soluble in water; soluble in alcohol
Heavy Metal (%) : 0.002%
Type : Other, Active Pharmaceutical Ingredient
Smell : Other, Odorless


 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free