Proguanil Hydrochloride IP
Price 4000 INR/ Kilograms
Proguanil Hydrochloride IP Specification
- Storage
- Store in tightly closed container, protected from light, at room temperature (15C to 25C)
- Poisonous
- No (when handled properly)
- Molecular Weight
- 290.19 g/mol Kilograms (kg)
- Shelf Life
- 3 years (when properly stored) Years
- Particle Size
- NMT 150 microns
- Structural Formula
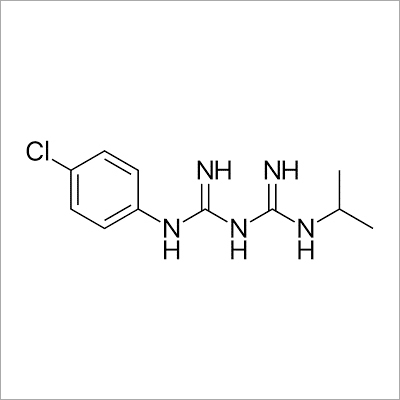
- C11H16ClN5HCl (see image for structure)
- Solubility
- Freely soluble in water, sparingly soluble in alcohol
- Taste
- Bitter
- Molecular Formula
- C11H16ClN5HCl
- HS Code
- 29339900
- Loss on Drying
- NMT 1.0%
- Smell
- Odorless
- Heavy Metal (%)
- NMT 0.001%
- Medicine Name
- Proguanil Hydrochloride IP
- Type
- API (Active Pharmaceutical Ingredient)
- Grade
- IP (Indian Pharmacopoeia)
- Usage
- Antimalarial drug intermediate; Used in the treatment and prophylaxis of malaria
- Purity(%)
- NLT 98.0% (by HPLC)
- Physical Form
- Solid
Proguanil Hydrochloride IP Trade Information
- Minimum Order Quantity
- 4 Kilograms
- Supply Ability
- 10000 Kilograms Per Month
- Delivery Time
- 15 Days
- Main Domestic Market
- All India
About Proguanil Hydrochloride IP
We have earned our name as a renowned organization engaged in offering Proguanil Hydrochloride IP Chemicals. These are biguanide compound, metabolize in the body to form cycloguanil, an anti-malaria agent. To comply with set safety norms, we use finest quality compounds, sourced from trusted vendors. These chemicals are active against several protozoal species and are used in combination with atovaquone and chloroquine for the prevention and therapy of malaria. Proguanil Hydrochloride IP Chemicals inhibit the bifunctional dihydrofolate reductase-thymidylate synthase (DHFR-TS) of plasmodium parasite. These chemicals are soluble in water; practically insoluble in chloroform and in ether.
Proguanil Hydrochloride IP
Name of Product: PROGUANIL HYDROCHLORIDEPROGUANIL HYDROCHLORIDE.jpg (12750 bytes)"
- Chloroguanide Hydrochloride
- Proguanil Hydrochloride is 1-(4-chlorophyll)-5- isopropyl biguanide hydrochloride.
- Formula: C11H16ClN5,HCl Category: Antimalarial.
- Mol. Wt.: 290.19
- Dose: Suppressive, 100 to 300 mg daily.
|
Analysis |
Specification |
|
Description |
White, crystalline powder; odorless. |
|
Solubility
|
Soluble in ethanol (95%); slightly soluble in water; practically insoluble in chloroform and in ether. |
|
Identification:
A:
D: |
By infra-red absorption spectrum
Gives the reactions of chlorides, |
|
Acidity or Alkalinity |
To 35 ml of water maintained at 60o to 65o add 0.2 ml of methyl red solution, neutralize with 0.01M sodium hydroxide or 0.01M hydrochloric acid, add 0.4 g of the substance being examined and stir until dissolved. The resulting solution is not acidic and requires for neutralization not more than 0.2 ml of 0.01M hydrochloric acid. |
|
Sulphates Ash
|
Not more than 0.1%
|
|
Loss on Drying |
Not more than 0.5%, determined on 1 g by drying in an oven at 105o |
|
Assay |
Proguanil Hydrochloride contains not less than 99.0 per cent and not more than 101.0 per cent of C11H16ClN5,HCl, calculated with reference to the dried substance. |
Indian Pharmacopoeia Grade Quality
Proguanil Hydrochloride IP is precisely manufactured to meet the stringent Indian Pharmacopoeia (IP) standards, ensuring reliable quality and consistent purity above 98%, verified by HPLC testing. Supplied as a fine solid with particle size not exceeding 150 microns, it is designed for pharmaceutical applications demanding accuracy and safety.
Application in Malaria Treatment
This API serves as an essential intermediate for antimalarial drugs, making it a crucial component in both preventive and therapeutic regimens for malaria. Its well-defined formulation supports consistent dosage and efficacious results, assisting manufacturers and healthcare professionals in achieving optimal therapeutic outcomes.
Safe Handling and Storage
Proguanil Hydrochloride IP is odorless and not considered poisonous when handled according to recommendations. For best results and maximum shelf life (3 years), it should be stored in tightly closed containers, away from light, and at controlled room temperatures between 15C and 25C.
FAQs of Proguanil Hydrochloride IP:
Q: How should Proguanil Hydrochloride IP be stored to maintain its quality?
A: Proguanil Hydrochloride IP should be stored in a tightly closed container, protected from direct light and maintained at room temperature between 15C and 25C. Proper storage ensures its stability and maximum shelf life of three years.Q: What is the primary use of Proguanil Hydrochloride IP in pharmaceuticals?
A: The main application of Proguanil Hydrochloride IP is as an intermediate in the formulation of antimalarial drugs, used both for prevention and treatment of malaria.Q: When is Proguanil Hydrochloride IP most beneficial for malaria prophylaxis?
A: Proguanil Hydrochloride IP is most beneficial as a preventative measure before exposure to malaria risk, as well as in ongoing treatment protocols, depending on healthcare guidelines and specific patient needs.Q: Where is Proguanil Hydrochloride IP manufactured and exported from?
A: This API is manufactured and supplied by exporters based in India, adhering to the Indian Pharmacopoeia standards for quality and purity.Q: What processes are involved in ensuring the purity of Proguanil Hydrochloride IP?
A: Purity is verified using high-performance liquid chromatography (HPLC), ensuring levels not less than 98%, alongside controls for loss on drying and heavy metals, making it suitable for pharmaceutical use.Q: How does the physical form and solubility aid in pharmaceutical formulation?
A: Being a solid with fine particle size and freely soluble in water, Proguanil Hydrochloride IP allows ease of processing, optimal dispersion, and reliable integration into liquid or solid dosage forms.Q: What are the benefits of using Proguanil Hydrochloride IP for manufacturers and end users?
A: Manufacturers benefit from its high purity, reliable quality, and compliance with IP standards, resulting in safe, effective malaria medications. End users receive trusted therapeutic products for malaria prevention and treatment.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Proguanil HCL Category
Proguanil Hydrochloride BP
Price 4000 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Molecular Weight : 290.20 g/mol Kilograms (kg)
Purity(%) : 98.0% Min.
Shelf Life : 3 years from date of manufacture Years
Heavy Metal (%) : Not more than 0.001%
Progunail Hydrochloride USP
Minimum Order Quantity : 5 Kilograms
Molecular Weight : 315.74 g/mol
Purity(%) : 99%
Shelf Life : 5 years
Heavy Metal (%) : 0.001%


 Send Inquiry
Send Inquiry

 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free