Progunail Hydrochloride USP
Progunail Hydrochloride USP Specification
- Molecular Formula
- C12H18ClN3O4
- Taste
- Bitter
- Smell
- Odorless
- Molecular Weight
- 315.74 g/mol
- Storage
- Store in a cool, dry place, away from light
- Loss on Drying
- 0.5%
- Particle Size
- Micronized; d90 < 50 microns
- EINECS No
- None reported
- HS Code
- 29420090
- Shelf Life
- 5 years
- Solubility
- Freely soluble in water and methanol
- Structural Formula
- Available on request
- Heavy Metal (%)
- 0.001%
- Type
- Active Pharmaceutical Ingredient (API)
- Grade
- USP
- Purity(%)
- 99%
- Physical Form
- Solid
- Related Substances
- Total impurities 0.2%
- Manufacturing Process
- Synthesized under GMP conditions
- Microbial Limit Tests
- Complies with Pharmacopeial standards
- Water Content
- Not more than 0.5%
- Packaging
- HDPE drums, 25 kg net
- Specific Optical Rotation
- +15 to +17
Progunail Hydrochloride USP Trade Information
- Minimum Order Quantity
- 5 Kilograms
- Supply Ability
- 10000 Kilograms Per Month
- Delivery Time
- 15 Days
- Main Export Market(s)
- Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Africa, Central America, Asia
- Main Domestic Market
- All India
About Progunail Hydrochloride USP
They are used for the causal prevention and suppression of malaria caused by susceptible strains. We have marked a distinct position in the domain by offering our clients Progunail Hydrochloride USP Chemicals. These are a prophylactic antimalarial drugs, work by stopping the malaria parasite. We process these chemicals at our sound processing unit using best quality compounds. The chemicals work by inhibiting the enzyme, dihydrofolate reductase, which is involved in the reproduction of the parasite. Progunail Hydrochloride USP Chemicals are combined with atovaquone for malaria prophylaxis. The two compounds act synergistically to inhibit the plasmodial dihydrofolate reductase (DHFR) and interrupt the electron transport chain.
FAQs of Progunail Hydrochloride USP:
Q: How is Progunail Hydrochloride USP manufactured to ensure high purity and compliance?
A: Progunail Hydrochloride USP is synthesized using advanced GMP (Good Manufacturing Practice) protocols to guarantee exceptional purity ( 99%) and compliance with USP standards. All manufacturing stages are closely monitored, including controls for water content, related substances, and microbial limits, to maintain pharmaceutical-grade quality.Q: What is the recommended storage condition and shelf life for Progunail Hydrochloride USP?
A: The product should be stored in a cool, dry place, away from light to retain its stability. When properly stored, Progunail Hydrochloride USP maintains its quality and effectiveness for up to five years from the date of manufacture.Q: When is this API most beneficial in pharmaceutical formulations?
A: Progunail Hydrochloride USP is most beneficial when a high-purity, freely soluble active ingredient is required for tablet, capsule, or injectable formulations. Its micronized particle size ensures uniformity and efficient absorption in various pharmaceutical applications.Q: Where is Progunail Hydrochloride USP manufactured and how is it supplied?
A: This API is manufactured in India by certified exporters and suppliers. It is securely packaged in 25 kg net HDPE drums that protect the product during transit and storage, ensuring it arrives in optimal condition.Q: What advantages does the micronized solid form of Progunail Hydrochloride provide?
A: The micronized solid form (d90 < 50 microns) enhances solubility and ensures uniform dispersion in formulations. This fine particle size improves the bioavailability of the final pharmaceutical product, supporting improved therapeutic outcomes.Q: How does the product meet microbial and heavy metal safety parameters?
A: Every batch undergoes rigorous testing according to pharmacopeial standards for microbial limits and contains heavy metal content not exceeding 0.001%. This ensures the product is safe for pharmaceutical use and minimizes risks associated with contamination.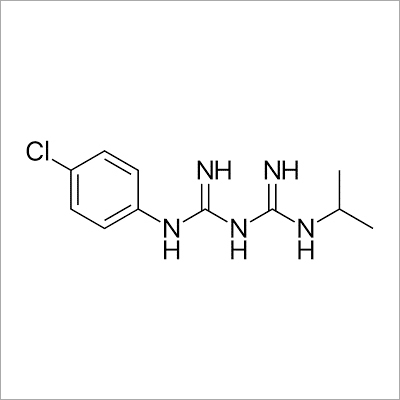

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Proguanil HCL Category
Proguanil Hydrochloride BP
Price 4000 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Physical Form : Solid
HS Code : 29332990
Grade : Other, BP (British Pharmacopoeia)
Type : Other, Active Pharmaceutical Ingredient
Proguanil Hydrochloride IP
Price 4000 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Physical Form : Solid
HS Code : 29339900
Grade : Other, IP (Indian Pharmacopoeia)
Type : Other, API (Active Pharmaceutical Ingredient)


 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free