Clomiphene Citrate BP
Price 7000 INR/ Kilograms
Clomiphene Citrate BP Specification
- Molecular Weight
- 598.09 g/mol Kilograms (kg)
- Boiling point
- Not available
- Residue on Ignition
- Not more than 0.2%
- Place of Origin
- India
- Melting Point
- 116118C
- Storage
- Keep container tightly closed in a cool and dry place, protected from light
- Moisture (%)
- Not more than 1.0%
- HS Code
- 29339900
- Loss on Drying
- Not more than 1.0%
- Heavy Metal (%)
- Not more than 0.001%
- Molecular Formula
- C26H28ClNOC6H8O7
- Other Names
- Clomifene citrate, Clomifene, Clomid, Serophene, Omifin
- Type
- Pharmaceutical Raw Material
- Grade
- Pharmaceutical Grade
- Usage
- Used to treat infertility in women
- Purity
- Not less than 98.0%
- Appearance
- White or almost white crystalline powder
- Application
- Other
- Raw Material
- Yes
- Smell
- Odorless
- Color
- White
- Form
- Powder
Clomiphene Citrate BP Trade Information
- Minimum Order Quantity
- 5 Kilograms
- Payment Terms
- Cash on Delivery (COD), Cheque, Cash Against Delivery (CAD), Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 10000 Kilograms Per Month
- Delivery Time
- 15 Days
- Main Domestic Market
- All India
About Clomiphene Citrate BP
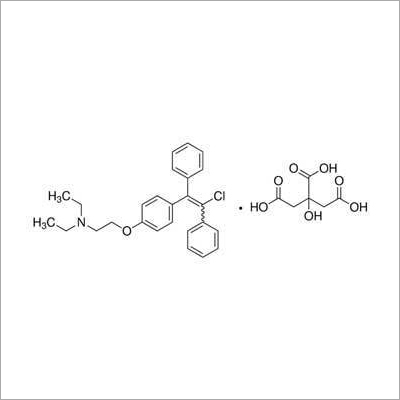
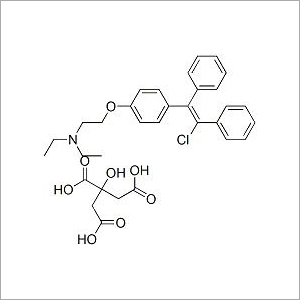
Name of Product: CLOMIPHENE CITRATE BP
Formula:C26H28ClNO,C6H8O7
Mol. Wt.: 598.1
|
Analysis |
Specification |
|
Description |
White or Pale yellow, crystalline powder |
|
Solubility |
Slightly soluble in water, sparingly soluble in alcohol |
|
Identification: A: B: |
By infra-red absorption spectrum A deep red Colour is produced. |
|
Water |
Not More than 1.0 % |
|
Related Substances |
Not more than 2.5 % ( Total Impurity) |
|
Z-ISOMER |
Between 30.00 % and 50.00 % |
|
Assay |
Clomiphene citrate contains not less than 98.0 per cent and not more than 101.0 per cent of calculated on the anhydrous substance. |
FAQs of Clomiphene Citrate BP:
Q: How is Clomiphene Citrate BP typically used in medical settings?
A: Clomiphene Citrate BP acts as a fertility drug and ovulation inducer, commonly prescribed for women facing infertility due to ovulatory dysfunction. It is formulated into tablets or capsules for oral administration under medical supervision.Q: What is the molecular composition and appearance of Clomiphene Citrate BP?
A: This raw material has a molecular formula of C26H28ClNOC6H8O7 and a molecular weight of 598.09 g/mol. It appears as a white or almost white, odorless, crystalline powder with a melting point between 116118C.Q: When and where should Clomiphene Citrate BP be stored to maintain quality?
A: Clomiphene Citrate BP should be stored in a cool, dry place, out of direct sunlight, with its container tightly closed. This preserves its stability and ensures it retains its high purity and effectiveness.Q: What steps are involved in the manufacturing and quality assurance process for this product?
A: Manufacturers in India follow stringent pharmaceutical protocols, including purity checks (not less than 98.0%), control of moisture content, residue on ignition, and heavy metals, along with visual and chemical analysis to ensure consistent pharmaceutical grade quality.Q: What are the benefits of using pharmaceutical grade Clomiphene Citrate BP?
A: The main benefit is effective induction of ovulation in women experiencing infertility. Its high purity and compliance with pharmaceutical standards minimize risk and maximize treatment efficacy, making it suitable for formulation into regulated fertility medications.Q: Where does this pharmaceutical raw material originate, and who supplies it?
A: Clomiphene Citrate BP is manufactured and exported by suppliers based in India, serving global pharmaceutical clients as a reliable source of quality-controlled raw material for fertility drugs.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Clomiphene Citrate Category
Clomiphene Citrate USP
Price 7000 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Type : Other, Active Pharmaceutical Ingredient
Color : Other, White to offwhite
Form : Powder
Heavy Metal (%) : Not more than 0.001%
Clomiphene Citrate IP
Price 4500 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Type : Other, Pharmaceutical Raw Material
Color : White
Form : Other,
Heavy Metal (%) : Not more than 0.001%


 Send Inquiry
Send Inquiry

 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free